
Corrective and Preventive action | CAPA management by Operon Strategist Medical Device Manufacturing Consultant - Issuu

Amazon.com: Principles of corrective Action and Preventive Action :CAPA: A Handbook for Quality Professionals in Medical device and Pharmaceutical Industries eBook : MUCHEMU, DAVID N.: Kindle Store

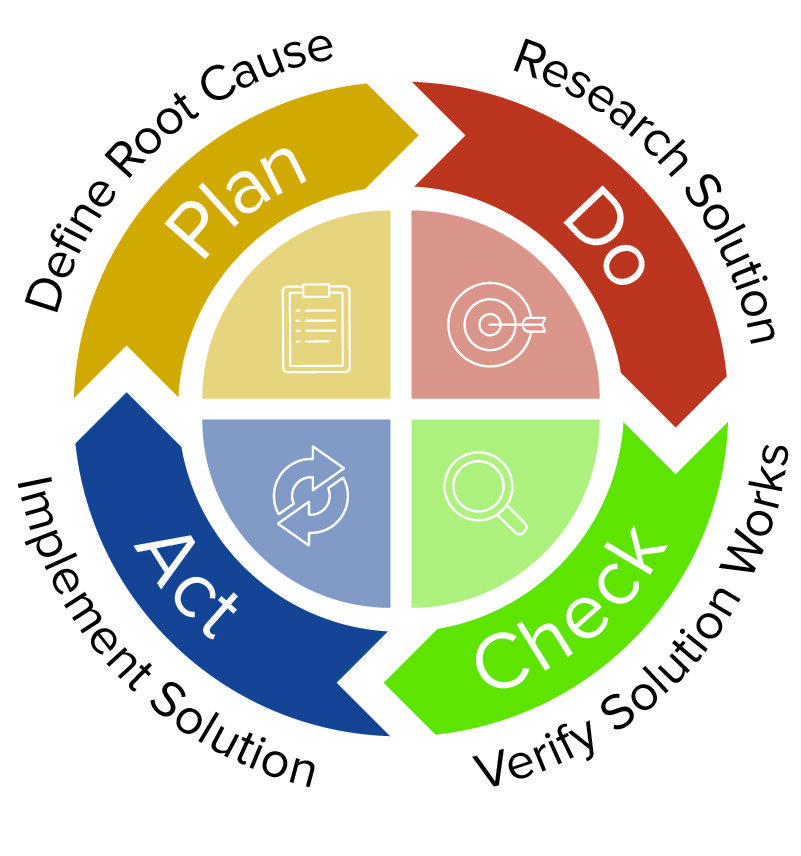

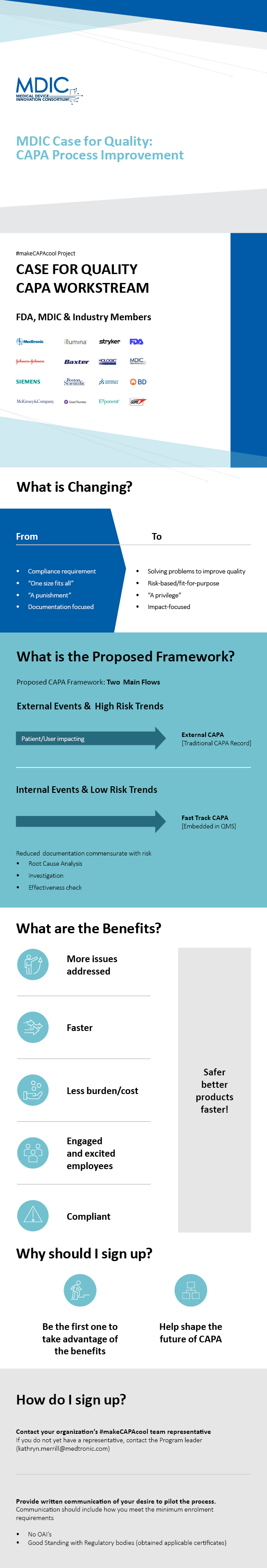
![Corrective and Preventive Action (CAPA): The Definitive Guide [2022] Corrective and Preventive Action (CAPA): The Definitive Guide [2022]](https://www.thefdagroup.com/hs-fs/hubfs/Screen%20Shot%202021-04-26%20at%2012.58.17%20PM.png?width=1229&name=Screen%20Shot%202021-04-26%20at%2012.58.17%20PM.png)









